Chronic heartburn isn’t just annoying-it can quietly change the lining of your esophagus in ways that put you at risk for cancer. This isn’t a rare edge case. About 5.6% of people in the U.S. have Barrett’s esophagus, a condition triggered by long-term GERD (gastroesophageal reflux disease). For many, it happens without warning. No sudden pain. No red flag. Just years of acid burning through the esophagus, slowly replacing normal tissue with abnormal, precancerous cells.
What Exactly Is Barrett’s Esophagus?
Barrett’s esophagus isn’t a disease on its own. It’s a physical change in your esophagus caused by repeated exposure to stomach acid. Normally, the esophagus is lined with flat, pink cells called squamous epithelium. When acid keeps hitting that area, those cells die off. The body tries to repair itself by growing new cells-ones that look more like the lining of the intestine. This is called intestinal metaplasia. It’s the hallmark of Barrett’s.
This change was first described in 1950 by Norman Barrett at St. Thomas’ Hospital in London. Since then, we’ve learned it’s not just a curiosity-it’s a warning sign. About 10-15% of people with chronic GERD develop it. And while only 5% of those with Barrett’s will ever get esophageal cancer, the cancer that does develop is aggressive. Less than 20% of people survive five years after diagnosis.
The timeline matters. It usually takes at least 10 years of frequent acid reflux before Barrett’s starts forming. And if you’ve had symptoms more than three times a week for over 20 years, your risk jumps 40 times higher than someone without GERD.
Who’s Most at Risk?
This isn’t an equal-opportunity condition. Men are three times more likely than women to develop Barrett’s esophagus. White men over 50 with long-term GERD are the highest-risk group. Obesity, especially belly fat, increases pressure on the stomach and worsens reflux. Smoking is another major trigger-it weakens the lower esophageal sphincter and reduces healing capacity.
Even if you’re not overweight or a smoker, long-term GERD alone puts you on the radar. The American College of Gastroenterology says screening should be considered for men with GERD lasting more than five years, especially if they’re over 50, white, and have other risk factors. Women and younger people rarely get screened unless they have multiple red flags.
Can You Feel It?
No. That’s the dangerous part. Barrett’s esophagus doesn’t cause its own symptoms. If you have it, you’re probably feeling the same old heartburn, regurgitation, or chest pain you’ve had for years. Some people report trouble swallowing solid food or waking up with a sour taste in their mouth. Others get chronic cough or hoarseness.
But here’s the trap: people normalize these symptoms. They think, “It’s just acid reflux. Everyone gets that.” So they don’t seek help until they’re in serious trouble. The Esophageal Cancer Action Network found that 68% of Barrett’s patients had symptoms for over five years before being diagnosed. By then, the damage may already be advanced.
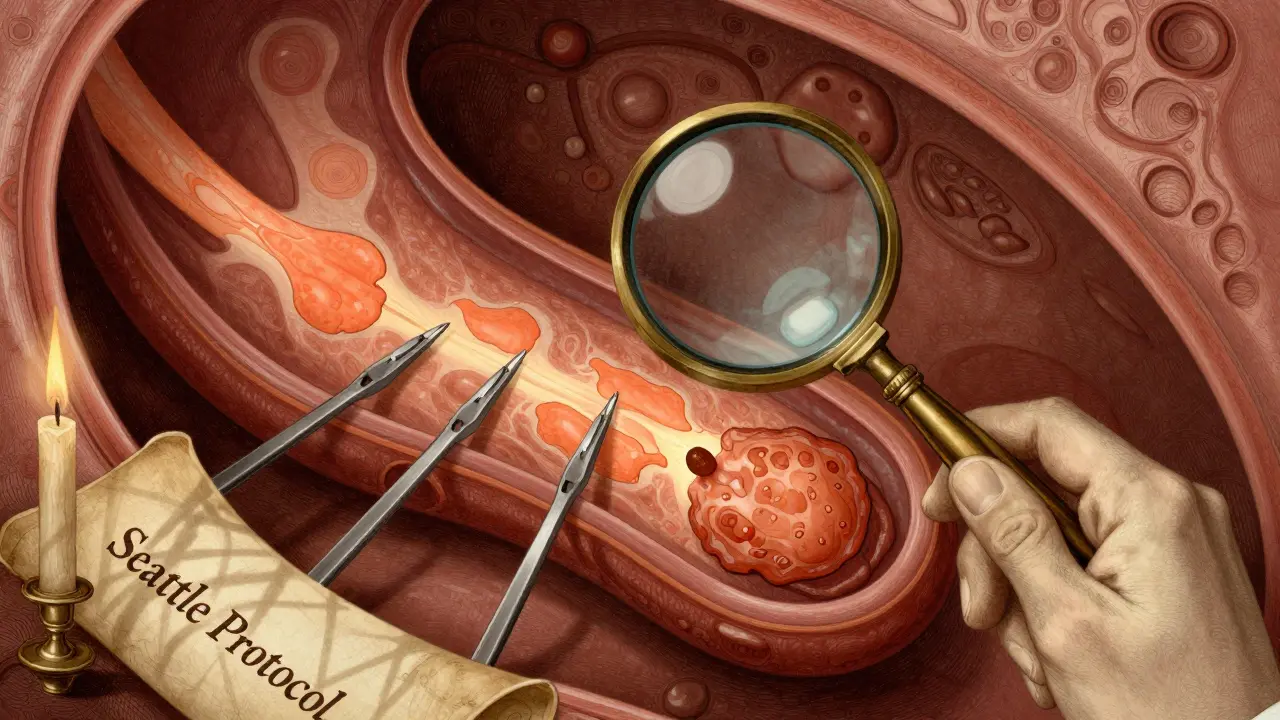
How Is It Diagnosed?
You can’t diagnose Barrett’s with a blood test or an X-ray. The only way is an upper endoscopy. A thin, flexible tube with a camera is inserted through your mouth into your esophagus. The doctor looks for abnormal tissue-typically a salmon-colored patch above the usual junction between the esophagus and stomach.
But seeing it isn’t enough. Biopsies are required. The standard method, called the Seattle protocol, means taking four tissue samples every 1 to 2 centimeters along the abnormal area. That’s usually 12 to 24 biopsies in one session. Why so many? Because dysplasia-the step before cancer-can be patchy. One missed spot could mean missing early warning signs.
The results are categorized:
- Non-dysplastic Barrett’s (NDBE): No abnormal cell changes. Most common.
- Indefinite for dysplasia: The pathologist isn’t sure. Needs repeat testing.
- Low-grade dysplasia (LGD): Early cell changes. Still not cancer, but closer.
- High-grade dysplasia (HGD): Severe changes. Nearly cancer. Requires immediate action.
Studies show HGD carries a 6-19% chance of turning into cancer each year. That’s why it’s treated aggressively.
Screening Guidelines: Who Gets Tested and When?
There’s no universal agreement on who should be screened. The American Gastroenterological Association recommends endoscopic screening for men with chronic GERD (more than 5 years) who also have at least one other risk factor: age over 50, white race, central obesity, or smoking history.
For women, screening is rarely recommended unless there’s a strong family history of esophageal cancer or multiple risk factors. The reason? Cost-effectiveness. Barrett’s is rare in women, and the number of procedures needed to prevent one cancer is too high to justify routine screening.
Once diagnosed, surveillance depends on the grade:
- NDBE: Repeat endoscopy every 3-5 years.
- LGD: Confirm with a second pathologist, then repeat endoscopy in 6-12 months. If stable, extend to every 2-3 years.
- HGD: Don’t wait. Treat it. Surveillance isn’t enough.
Many patients report confusion here. One Reddit user wrote: “Three different gastroenterologists gave me three different schedules for my non-dysplastic Barrett’s.” That’s not unusual. Guidelines vary, and doctors interpret them differently.
Treatment: Beyond Just Pills
Taking a proton pump inhibitor (PPI) like omeprazole doesn’t fix Barrett’s. It may reduce your heartburn, but studies show only 55-70% of patients get full acid suppression-even on high doses. That means the esophagus is still getting hit with acid, even if you don’t feel it.
True management requires two things: complete acid control and surveillance.
Lifestyle changes matter:
- Avoid fatty foods, chocolate, caffeine, and spicy meals.
- Don’t eat within 3 hours of lying down.
- Elevate the head of your bed 6-8 inches.
- Maintain a BMI under 25.
For patients with dysplasia, especially HGD, endoscopic ablation is now standard. Techniques like radiofrequency ablation (RFA) and cryotherapy can remove the abnormal tissue. Studies show RFA eradicates dysplasia in 90-98% of cases. One 2010 trial found 77.4% of patients had complete removal of abnormal cells after one year. Since then, RFA has become the go-to treatment.
In 2022, guidelines expanded RFA use to include all confirmed cases of low-grade dysplasia. The AIMS-2 trial showed 94% of LGD patients had durable eradication after five years. That means fewer people need lifelong surveillance.
The Big Problem: Too Many Procedures, Not Enough Answers
Here’s the uncomfortable truth: 95% of people with Barrett’s esophagus will never develop cancer. But we can’t tell who will. So we screen everyone. We do endoscopies. We take biopsies. We burn away tissue. It’s expensive-over $1.2 billion a year in the U.S. alone.
Dr. Stuart Spechler, writing in the New England Journal of Medicine, put it bluntly: “The fundamental challenge remains identifying which of the 5.6% of Americans with Barrett’s esophagus will progress to cancer.”
That’s why new tools are emerging. The TissueCypher Barrett’s Esophagus Assay is a non-endoscopic test that analyzes molecular markers in esophageal cells. It got Medicare coverage in 2021 after a study showed a 96% negative predictive value-meaning if the test says you’re low risk, you almost certainly are. This could cut unnecessary endoscopies by 40%.
Researchers are also testing DNA methylation markers. A $2.4 million study running from 2023 to 2026 is looking at whether these biomarkers can predict progression better than current methods. If they work, we might soon have a blood test or saliva test to identify high-risk patients.
What Should You Do?
If you’ve had GERD for more than five years, especially if you’re a man over 50, white, overweight, or a smoker: talk to your doctor about screening. Don’t wait until your symptoms get worse. The damage is already happening.
If you’ve been diagnosed with Barrett’s:
- Stick to your surveillance schedule. Don’t skip endoscopies.
- Take your PPIs as prescribed-but know they’re not a cure.
- Make lifestyle changes. Weight loss and sleep posture matter more than you think.
- Ask about ablation if you have dysplasia. It’s safe, effective, and often curative.
And if you’re unsure whether you need screening? Ask: “Based on my history, am I in the high-risk group?” Don’t assume you’re fine just because you don’t feel sick. Barrett’s doesn’t shout. It whispers. And by the time it yells, it’s often too late.
Can Barrett’s esophagus go away on its own?
No. Once the esophagus has developed intestinal metaplasia, the change is permanent unless treated. Endoscopic ablation can remove the abnormal tissue, and in many cases, the normal lining regrows. But without treatment, Barrett’s doesn’t reverse itself. Even if symptoms improve with PPIs, the underlying cell change remains.
Do all people with GERD get Barrett’s esophagus?
No. Only about 10-15% of people with chronic GERD develop Barrett’s esophagus. But that still means millions of people are at risk. The key factors are duration and frequency of reflux. If you’ve had heartburn more than three times a week for over 20 years, your risk is much higher than someone who gets it once a month.
Is Barrett’s esophagus the same as esophageal cancer?
No. Barrett’s esophagus is a precancerous condition. It increases your risk of developing esophageal adenocarcinoma, but most people with Barrett’s never get cancer. The progression from Barrett’s to cancer usually takes years and passes through stages: metaplasia → low-grade dysplasia → high-grade dysplasia → cancer. Surveillance helps catch it early.
Can I skip endoscopies if I feel fine?
No. Barrett’s esophagus doesn’t cause new symptoms. Feeling fine doesn’t mean the tissue isn’t changing. Many patients are diagnosed with cancer during their first endoscopy because they never had symptoms beyond routine heartburn. Skipping surveillance is one of the biggest mistakes people make.
Are there alternatives to endoscopy for screening?
Yes, but they’re not yet standard. The TissueCypher test is a non-endoscopic option that analyzes molecular markers from a swallowable capsule. It’s approved for Medicare patients and has a 96% accuracy in ruling out high-risk changes. Other tests are in development, including blood and breath biomarkers. But endoscopy with biopsy is still the gold standard for diagnosis and monitoring.
What’s Next?
The future of Barrett’s esophagus management is moving away from one-size-fits-all surveillance. We’re heading toward personalized risk assessment-using genetics, molecular markers, and AI to identify who truly needs frequent endoscopies and who doesn’t. Until then, the best defense is awareness, timely screening, and aggressive management for those at risk. Don’t ignore chronic reflux. It’s not just a nuisance. It’s a silent signal.

Martin Halpin
February 27, 2026 AT 19:40Okay, but let’s be real-how many of these 'screening guidelines' are just there so hospitals can bill more endoscopies? I’ve got a buddy in Dublin who got flagged for 'non-dysplastic Barrett’s' after one weird heartburn episode, and now he’s got a yearly appointment like it’s a damn gym membership. Meanwhile, his cousin in Nigeria who’s had acid reflux since 1998? No screening, no endoscopy, just turmeric tea and prayer. Who’s really getting helped here? I’m not saying skip screening, but let’s stop pretending this isn’t a profit pipeline disguised as prevention.
And don’t get me started on the Seattle protocol. Twelve biopsies? That’s not medicine, that’s a medieval torture ritual with a camera. I’d rather get a CT scan and a DNA spit test than have my throat ripped open for the third time this year. Give me a blood test that doesn’t require me to fast for 12 hours and cry into a paper cup.
Eimear Gilroy
February 28, 2026 AT 12:16This was actually really well-explained. I’ve had GERD for over 15 years and never knew Barrett’s could develop without worsening symptoms. I always thought if I just took my omeprazole and avoided spicy food, I was fine. Turns out, my body’s been quietly remodeling my esophagus like a DIY project gone wrong.
I’m going to ask my GI about screening next visit. I’m 48, female, overweight, and have been on PPIs since 2010. I didn’t realize I was in the risk group-even though I’m not white or male. Maybe I should’ve been screened years ago. Thanks for the clarity.
Michael FItzpatrick
March 1, 2026 AT 14:13Man, this post reads like a TED Talk written by a gastroenterologist who’s seen too many late-stage cancers. I love it. Let me throw some real talk in here: the reason we’re doing all these endoscopies isn’t because we’re paranoid-it’s because we’re *reactive*. We waited too long to find a better way.
But here’s the silver lining: we’re finally moving toward precision medicine. TissueCypher? That’s the future. Imagine a world where you swallow a pill, get a report in 72 hours, and if you’re low-risk? You’re off the hook for five years. No scope. No biopsies. No anxiety. Just peace.
And for those of you still thinking ‘I feel fine, so I’m fine’-you’re not a patient, you’re a walking time bomb. Barrett’s doesn’t scream. It whispers. And by the time you hear it, your esophagus is already half-soup.
Do the work. Get screened. If you’ve got dysplasia? Don’t wait. Get ablated. RFA is basically magic. It’s like a laser tattoo removal… but for your insides. And yeah, it’s weird. But it works.
Brandice Valentino
March 1, 2026 AT 15:01Larry Zerpa
March 2, 2026 AT 22:46Let’s dismantle this narrative. You say ‘only 5% of Barrett’s patients develop cancer’-but you’re ignoring the fact that 100% of esophageal adenocarcinomas arise from Barrett’s. That’s not a coincidence. It’s a pathway. And you’re acting like this is some statistical footnote when it’s literally the only route to this deadly cancer.
And the ‘cost-effectiveness’ argument for not screening women? That’s not medicine-that’s sexism wrapped in a spreadsheet. If you’re a woman over 50 with 20 years of reflux, you’re not ‘low risk.’ You’re just statistically invisible. The data doesn’t lie-your body does. And if you’re not getting screened because of gender, you’re being abandoned by the system.
Also, ‘PPIs don’t fix Barrett’s’? No, they don’t. But they’re not useless. They’re the only thing keeping your esophagus from turning into a charred ruin. Stop pretending lifestyle changes alone are a cure. They’re not. They’re damage control. And if you think you can ‘eat less fat’ and ‘elevate your bed’ and magically reverse metaplasia, you’re living in a wellness influencer fantasy.
Gwen Vincent
March 4, 2026 AT 21:06I just wanted to say thank you for writing this with such care. I’ve been avoiding my GI for months because I was scared of what they might find. But reading this made me feel less alone. I’ve had GERD since college, I’m 42, and I’ve always felt like I was ‘overreacting’ when I mentioned my symptoms.
Now I’m scheduling my endoscopy. Not because I’m terrified-but because I want to know. Knowledge isn’t fear. It’s power. And if I have Barrett’s? I’ll get it treated. If I don’t? I’ll sleep better. Either way, I win.
To anyone else out there who’s been ignoring their reflux because ‘everyone gets it’-you’re not being tough. You’re being vulnerable. And that’s okay. Reach out. Get checked. You deserve to be safe.
Valerie Letourneau
March 6, 2026 AT 04:02While I appreciate the thoroughness of this exposition, I must respectfully note that the emphasis on demographic risk factors-particularly race and gender-may inadvertently reinforce systemic biases in clinical practice. The assertion that ‘women rarely get screened unless there are multiple red flags’ reflects a historical data gap, not a biological truth.
As a Canadian clinician who has worked in both public and private systems, I have observed that women with long-standing GERD are often dismissed as ‘anxious’ or ‘overly sensitive,’ leading to diagnostic delays. The current guidelines, while statistically grounded, may be perpetuating inequity under the guise of cost-efficiency.
Perhaps the future lies not in refining who gets screened, but in universal access to non-invasive biomarkers. Until then, we must advocate for individualized risk assessment, not population-based exclusion.
Khaya Street
March 6, 2026 AT 23:09Look, I’ve had acid reflux since I was 19. I’m 52 now. I’ve never been to a specialist. I drink ginger tea, sleep on three pillows, and take Tums like candy. This post scared me a little, but I’m not getting an endoscopy. Why? Because I don’t have the time. I work two jobs. My insurance is a joke. And I’ve got two kids in college.
So yeah, maybe I’m ‘at risk.’ But you telling me I need to take time off, drive 90 minutes, and pay $500 for a scope so some doc can cut out tissue I didn’t even know I had? Nah. I’ll keep living. If I get cancer? I get cancer. At least I lived my life without being scared by a blog post.
Christina VanOsdol
March 8, 2026 AT 11:05Brooke Exley
March 9, 2026 AT 04:35Hey everyone-I’ve been where you are. I was diagnosed with HGD two years ago. I was terrified. I thought I was going to die. But I got RFA. One session. No hospital stay. Went home the same day. A year later? My esophagus looks like new. Normal tissue. No dysplasia. Zero cancer.
It’s not a magic bullet, but it’s the closest thing we have. And if you’ve got dysplasia? Don’t wait. Don’t second-guess. Don’t listen to the ‘maybe it’ll go away’ people. That’s not hope-that’s denial.
You’re not alone. I’m here. I’ve been through it. And I’m still here. You can be too. Reach out. Ask questions. Get help. You’ve got this. 💪❤️